Testing Services
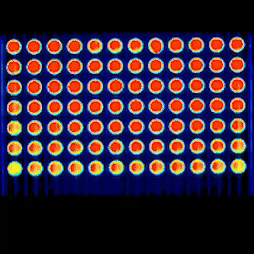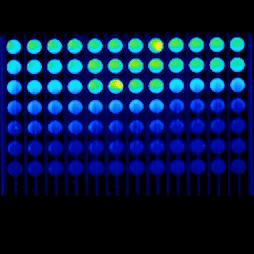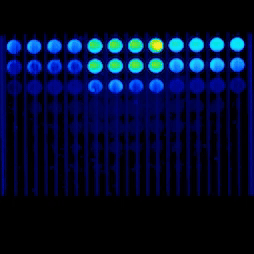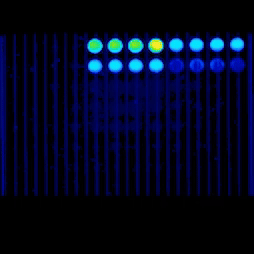
Our capabilities include viability and toxicity assays, high-content fluorescence readouts, flow cytometry analysis, and advanced microscopy, including confocal and live-cell imaging, to study cell health, death pathways, and dose–response effects.
Real-time kinetic plate imaging enables compound addition directly onto live cells with simultaneous monitoring of cellular responses across all wells. High-temporal-resolution measurements, acquired at sub-second intervals up to 120 measurements per second, allow detailed assessment of dynamic and transient cytotoxic effects that are not captured by endpoint assays alone.